We now have more information to get an idea how the process is performing. Each landing position is different and the variation may be due to common causes which are always present in this process. Look at the display of Landing positions at the right of the launcher. There is always a certain amount of variation in every process and if we only have common cause variation and we are trying to adjust for this variation we will actually cause more variation on the output. The answer is NO because we don’t know the process yet. The customer of this process wants the landing position to be 500, so what do we do now? Should we compensate for the error by moving the launcher? When we fire one ball from the launcher we find the value is 415. Your job is to fire balls at the target and get them to land as close as possible to the ideal value of 500. Imagine that you are the operator of a machine – the launcher. The location of the previous shots fired are given in the screen. The target is 500 and the specifications are 300 to 700. The process is a tennis ball launcher and we are trying to shoot balls at a target distance. Our job is to try to get results as close as possible to that target. In this lesson we are going to look at a process where we have been given a preferred target value for a variable measurement.
Attribute data, on the other hand, can only have whole number values like 1, 3, 12 etc. With variable data we can measure to any accuracy that we want, for example 12.5, 3.075 etc. The number of blemishes on a surface, the number of faulty products and the number of unpaid invoices. Length, weight, temperature and pressure.Īttribute data is based on discrete counts. Variable data is any measurement which has a continuous scale. Data can be divided into two major categories, variables and attributes. We use different types of control charts for different types of data.
XBAR CHART MINITAB HOW TO
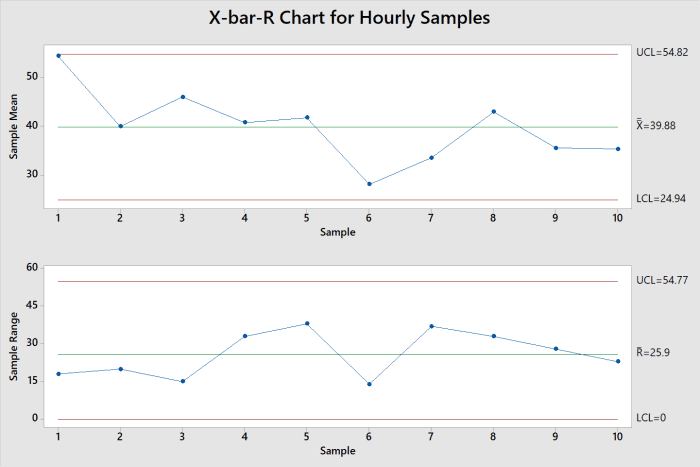
Now we are going to learn how to draw the control charts. This work provides guidance on identifying and setting default health‐based exposure limits (HBELs) for defined non‐hazardous substances in pharmaceutical manufacturing in line with regulatory guidelines.In lesson 1 we discovered why we need control charts. Contract Development and Manufacturing Organizations (CDMOs) work with a large, diverse portfolio of pharmaceutical substances, thus, often need a pragmatic approach to apply risk‐based decisions for patient and worker safety during manufacturing in multi‐purpose facilities. The proposed default limit values are 1 mg/m3 for the OEL, and 50 mg/day for the PDE oral and IV (intravenous) route. The non‐hazardous substances discussed within this publication are part of the following subgroups: (I) inactive pharmaceutical ingredients, (II) pharmaceutical excipients or cosmetic ingredients, (III) substances Generally Recognized as Safe (GRAS), and (IV) food ingredients, additives and contact materials. Since there is no aligned definition provided by authorities, nor agreed default values for non‐hazardous substances, we provide a decision tree in order to help qualified experts (such as qualified toxicologists) to identify the group of non‐hazardous substances and to assign default HBEL values for specific routes of exposure. While pharmaceuticals require the establishment of health‐based exposure limits (HBELs) (e.g., occupational exposure limits, permitted daily exposure limits), the limits for non‐hazardous substances could be set in a protective and pragmatic way by using default values, when internally required. Additionally, CDMOs may not have access to primary study data or data is generally lacking for a specific substance. Contract Development and Manufacturing Organizations (CDMOs) that manufacture large, diverse portfolio of chemical and pharmaceutical substances require pragmatic risk‐based decisions with respect to the safe carry‐over between different chemical entities, as well as for worker protection.
